|
|
ТОМ 4, СТ. X (сc. X) // Апрель, 2003 г.
ACUTE POISONINGS BY NEW PSYCHOTROPIC SUBSTANCE ZOPICLONE L. Khodasevitch Latvian State Center for Forensic Medical Examinations Riga Republic of Latvia Introduction
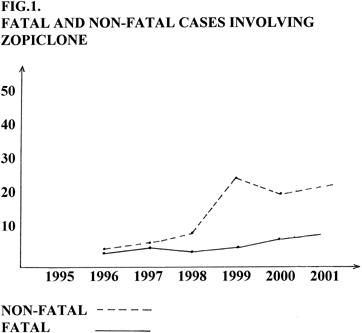
Zopiclone is the third generation non-benzodiazepine hypnotic agent, that chemically belongs to the cyclopyrrolones. Zopiclone appeared in France in 1987, but in Latvia-since 1995. Zopiclone is widely used and abused in Latvia. It could be explained by its popularity and availability in drug stores without prescription. Zopiclone poisonings became a serious problem in Latvia.From 1996 more than 60 cases involving zopiclone (both fatal and non-fatal) were registered in Latvia (Fig. 1). Signs of zopiclone acute overdose are CNS depression, respiratory depression, coma, hyperkalemia, hyperglycemia. No specific signs of zopiclone intoxication are seen at the autopsy. So results of toxicological analysis are of great importance. Post-mortem blood levels associated with fatal overdoses of zopiclone are 1,4-3,9 mg/L. Van Bocxlaer et al. have described a case of poisoning by zopiclone and found drug concentrations of 0,15 mg/kg in stomach contents, 5,10 mg/kg in liver and 1,18 mg/L in blood [1]. In similar fatality case [2] concentrations ranging from 0,9 to 2,0 mg/L in ten distinct blood sampling sites were found.Post-mortem blood levels of zopiclone are relatively stable and little influenced by post-mortem redistribution [3], which is consistent with the known low volume of distribution, approximately 1,5 L/kg [4]. The highest drug concentrations were found in spleen (5,8 mg/kg), but this may reflect post-mortem diffusion from stomach contents (55,1 mg/L). Liver concentrations measured up to 4,9 mg/kg, but they were site variable and ranged between 0,5 and 4,9 mg/kg in eight samples. The highest concentrations were in the left lobe, and the lowest con centration in the right lobe [5]. A 29-year-old female weighind 64 kg had cardiac blood ethanol of 1,53‰ and zopiclone blood concentration 3,0 mg/L [2]. Post-mortem blood femoral concentrations were 0,03-0,4mg/L [7]. P. Boniface et al. described two cases of fatal zopiclone poisoning. Quantitative determinations of zopiclone yielded 1,4-3,9 mg/L in the blood, 0,81 and 8,7 mg/kg in liver and 13,5 and 133 mg/kg in stomach contents [8]. A fatality, attributed to a suicidal ingestion of 450 mg of zopiclone and subsequent death by drowning was described [9].
Figure 1. Fatal and non-fatal cases involving zopiclone.
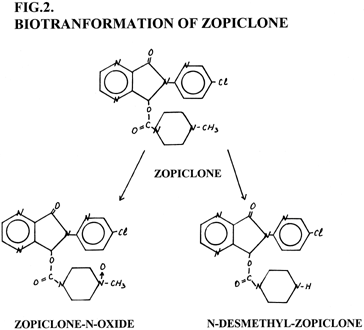
Therapeutic plasma concentrations of zopiclone vary from 10 to 100 mkg/L. Zopiclone is white, odourless and non-hygroscopic powder, practically insoluble in water and ethanol. Several methods have been described for zopiclone detection in plasma and urine for pharmacokinetics and biotransformation studies. Basic pharmacokinetic data of zopiclone (Tab. 1) and biotransformation (Fig. 2).
Figure 2. Biotransformation of zopiclone. The elimination half-life of zopiclone is short (4-5 h.), 31% of the dose is recovered by urinary excretion, of which only 5% is represented by unchanged drug,with N-desmethyl-zopiclone being the most important fraction (15%) and zopiclone N-oxide accounting for 11% of the dose. Zopiclone metabolites have similar UV-spectral, TLC- and GC-features (Tab. 2, 3). Our experiments showed that zopiclone is unstable at high temperature and in alkaline. The main task of our study was to create simple method for zopiclone analysis in blood and urine by means GC-FID and GC-ECD.
Table 1. Basic pharmacokinetic data of zopiclone. Proposed single oral dose (mg) 7.5 Bioavailability: oral (%) 80 Absorption within 1.5 h Volume of distribution (L/kg) 1.5 Plasma protein binding (%) 45 Elimination half-life (h) 5 Renal excretion of unchanged substance in urine (%) 2-4 Renal plasma clearance (L/h) 14
Table 2. UV-maxima of zopiclone and metabolites. SUBSTANCE UV-MAXIMA (nm) Methanol 0.1N HCl 0.1N NaOH ZOPICLONE 304 303 237 277 N-OXIDE- 304 304 237 277 DESMETHYL- 304 304 237 277
Table 3. GC retention indices of zopiclone and metabolites SUBSTANCE RETENTION INDEX ZOPICLONE 3051 N-OXIDE- 3050 DESMETHYL- 3089
Materials and methods
Blood and urine samples in cases of fatal and non-fatal intoxications Methods of TLC,GC-FID and GC-ECD were used after alkaline hydrolysis of specimens as described in [9]. 2-amino-5-chloropyridine (Aldrich, CAS 1072-98-6) was used. Results and discussion
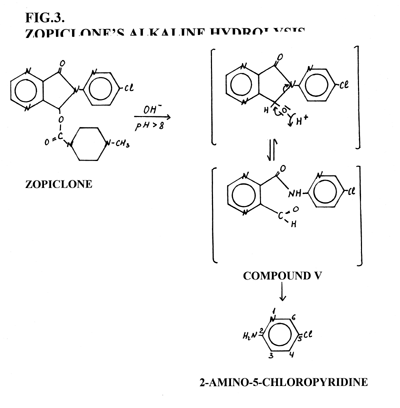
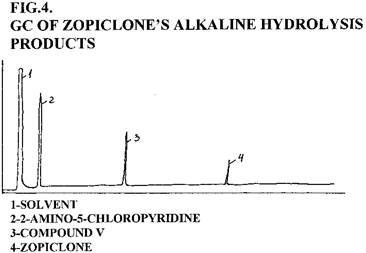
During alkaline hydrolysis 2-amino-5-chloropyridine was identified on the TLC plates as a violet spot with Fast Black K reagent, and by means GC-FID and GC-ECD. Alkaline hydrolysis products (Fig. 3) usually include 2-amino-5-chloropyridine, compound V and a small amount of zopiclone. Zopiclone and its alkaline hydrolysis products have different retention times on GC chromatogram (Fig. 4) and Rf values on the TLC plates (Tab. 4.) and could be successfully identified.
Figure 3. Zopiclone's alkaline hydrolysis products.
Figure 4 . GC of zopiclone's alkaline hydrolysis products. Table 4. TLC data of zopiclone and its hydrolysis products SUBSTANCE Rf I II III ZOPICLONE 0.34 0.29 0.41 COMPOUND V 0.63 0.58 0.68 2-AMINO-5-CHLORO- PYRIDINE 0.95 0.89 0.93 I-BENZENE:1,4-DIOXANE:25%AMMONIUM HYDROXIDE (60:35:5) II-ETHYL ACETATE:METHANOL:25%AMMONIUM HYDROXIDE (17:2:1) III-METHANOL:25%AMMONIUM HYDROXIDE (100:1.5)
References
|
||||||||||||||
| Размещение рекламы | |
|
| |

 Articles
Articles