|
|
ТОМ 4, СТ. 77 (стр. 113-119) // Март, 2003 г.
EXPERIMENTAL OBSERVATIONS RELATED TO THE UTILITY OF MELATONIN IN ATTENUATING AGE-RELATED DISEASES "Успехи геронтологии", 1999г., выпуск 3 Russel J. Reiter Department of Cellular and Structural Biology The University of Texas Health Science Center 7703 Floyd Curl Drive San Antonio, TX 78284-7762 Phone: 210/567-3859 Fax: 210/567-6948 E-mail: reiter@uthscsa.edu
Running title: Melatonin and age-related diseases
For: Advances in Gerontology Abstract. Melatonin was recently discovered to be free radical scavenger; in this capacity it very effectively detoxifies the most highly toxic oxygen-based radical, that is, the hydroxyl radical, while also directly neutralizing nitric oxide, the peroxynitrite anion, singlet oxygen, and to some degree the peroxyl radical. Besides its direct free radical scavenging activities, melatonin also may stimulate the activity of several antioxidative enzymes including the superoxide dismutases, glutathione peroxidase, glutathione reductase, and glucose-6-phosphate dehydrogenase. Conversely it inhibits neural nitric oxide synthase, a pro-oxidative enzyme. In both in vitro and in vivo studies melatonin has been shown to reduce oxidative damage to membrane lipids, proteins and nuclear DNA. It readily crosses all morphophysiological barriers and gets into every cell with ease. The bulk of the studies performed to date have used what seem to be pharmacological levels of melatonin to achieve antioxidative protection, but there is also evidence that physiological melatonin concentrations contribute to the total antioxidant status of the organism. In model systems of age-related diseases (cancer, cataracts, Alzheimer's disease, Huntington's disease, Parkinsonism) melatonin has been shown to be protective. Melatonin levels diminish with age in mammals including man. The association between the age-related reduction in melatonin and the onset of free radical-related diseases in the aged is currently being investigated.
Aging is the accumulation of adverse changes which lead to functional deterioration and increase the chances of death (47). These changes are generally considered responsible for a wide variety of diseases, which typically occur after middle age and often result in the death of an individual. The physiological alterations that accompany aging are attributable to a variety of factors including genetic, developmental, environmental, and the inborn processes referred to as aging (39).
Many theories have been advanced to explain the process of aging and their contribution to age-associated diseases. While no one theory has proven to be the definitive explanation for aging, one that has received a great deal of attention is the free radical theory (38, 39, 47). The free radical theory states that aging and the associated diseases are a consequence of the life long destruction and accumulation of essential macromolecules damaged by toxic free radicals. A corollary of this theory is that administration of molecules that either prevent the formation of radicals or neutralize them once they are formed would defer the rate of aging and reduce the incidence of age-associated diseases (44, 46, 94, 96, 97). To date, studies carried out to achieve this result have met with limited success. Molecules which reduce the damage inflicted by radicals are referred to as free radical scavengers and antioxidants of which there are many (68, 111).
One newly discovered agent that is a highly effective antioxidant and which has already been widely tested for its ability to protect against free radical-related processes is N-acetyl-5-methoxytryptamine, commonly known as melatonin (36, 37, 86, 95, 105). The purpose of this review is to summarize what is known about melatonin as a free radical scavenger and antioxidant and to discuss these findings relative to the free radical theory of aging.
Melatonin as a Free Radical Scavenger
Many free radicals are derived from the use of molecular oxygen (O2) by aerobic organisms. While roughly 97% of the O2 inhaled is utilized by mitochondria for the generation of energy, i.e., ATP, the remainder is reduced to free radicals and reactive oxygen intermediates (ROI) which have varying degrees of toxicity (Fig. 1) (24, 35).
By definition a free radical is a species that has an unpaired electron
(e-) in its outer orbital. Since e- are
normally paired in molecules, this feature makes the molecules that
possess them highly reactive (19). ROI on the other hand, do not
possess an unpaired e- but they may, however, be highly
reactive and, furthermore, when metabolized they generate free
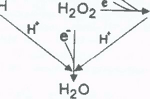
radicals. One of the most widely investigated ROI is H2O2;
its importance stems from its metabolism to the most reactive and
toxic free radical, the hydroxyl radical (·OH)
(57). The ·OH is generally
considered the most damaging of the oxygen-based free radicals that
are produced and it is believed to account for an estimated 50% of
the total damage induced by free radical mechanisms (42). Thus, any
molecule that would efficiently scavenge the ·OH
and importantly be in the vincinity of where ·OH
is produced intracellularly would be considered an important
antioxidant. The importance of the free radical scavenger being at
the site where the ·OH is
generated is important since, due to its very high reactivity, the
·OH travels no more than
several Angstroms before it interacts with another molecule.
This distance has been referred to as the reaction age of the ·OH
and a free radical scavenger must be within this "cage"
to neutralize the radical (15). To put this in the context of a
cell, if an antioxidant is confined to the lipid-rich membrane of a
cell because of its specific solubility, it will be ineffective in
reducing ·OH damage to DNA
in the nucleus.
In 1993, it was shown that melatonin is highly efficient ·OH scavenger in vitro (117). The definitive test to verify the ability of a free radical scavenger to neutralize a radical is considered to be the identification of the end product by electron spin resonance spectroscopy (ESR). Tan and colleagues (117) used ESR to verify that melatonin reduced adduct formation, in this case DMPO-·OH, in the following reaction:
Normally, the exposure of hydrogen peroxide (H2O2) to 254 nm ultraviolet light causes the formation of the ·OH. However, ·OH rapidly interact with each other because they each possess an unpaired e- in what is referred to as termination reactions. Thus, a spin trapping agent, in this case 5-5-dimethyl-pyrroline N-oxide or DMPO, was added. The spin trap forms an adduct with the generated ·OH (DMPO-·OH) which is quantified by ESR (90). The number of DMPO-·OH adducts formed is used as an index of ·OH generation. In this scheme, the addition of melatonin to the reaction mixture reduced the formation of the DMPO-·OH adducts illustrating the indole scavenged the ·OH (117). Furthermore, this group showed that melatonin was more effective than either reduced glutathione, an endogenously synthesized ·OH radical scavenger, or mannitol, an exogenously produced ·OH scavenger, in reducing DMPO-·OH adduct formation.
These seminal observations led to a series of studies in which the ·OH scavenging activity of melatonin was confirmed. Thus, also using ESR technology, Matuszek et al. (58) and Susa and colleagues (116) re-affirmed the high efficiency of melatonin as a ·OH radical scavenger. Likewise, Stasica and co-workers (114) also reported that melatonin reduces ·OH generation in vitro. That indoles with a structure similar to melatonin may also have free radical scavenging activity, although less than that of melatonin, was demonstrated in the studies by Tan and co-workers (117), Poeggeler et al. (85) and Matuszek et al. (58) (Table 1). The ability of melatonin to scavenge the ·OH was also shown by Pahkla et al. (73) who used yet another endpoint to estimate the ·OH scavenging activity of melatonin. Thus, they used terephthalic acid (THA) as a chemical index of ·OH formation since it forms an adduct, i.e., THA-·OH, which can be quantified. Melatonin, in a concentration-dependent manner, inhibited the formation of THA-·OH indicating reduced ·OH availability. In this system the concentration of melatonin required to inhibit ·OH formation by 50%, i.e., the IC50, was 11.4 +/- 1.0 µM (73). This is similar to the IC50 value (21 µM) reported by Tan et al. (117) where melatonin was found to reduce DMPO-·OH adduct formation.
Melatonin has also been shown to be a powerful inhibitor of ·OH-mediated macromolecular damage (94, 103, 104). Hence, H2O2 toxicity, which is widely accepted to be a consequence of the conversion of H2O2 to ·OH (61), is readily suppressed by the addition of melatonin to tissue homogenates (25, 109, 112). This is consistent with the observation of Li and co-workers (50) who showed that, in vivo, melatonin scavenges the ·OH as indicated by melatonin's suppression of dihydrobenzoic acid (DHBA) in the microdialysate collected from brains undergoing ischemia.
Even more definitive proof of melatonin's in vivo ·OH scavenging activity was provided by the recent observation that the administration of melatonin to animals subjected to ionizing radiation increased the amount of cyclic 3-hydroxymelatonin in their urine (118). Using a combination of mass spectrometry (MS), proton nuclear magnetic resonance (lH-NMR), thermodynamic stability studies and high performance liquid chromatography with electrochemical detection (HPLC-EL), we showed cyclic 3-hydroxymelatonin to be an end product of the interaction of melatonin with two hydroxyl radicals (Fig. 2). This end product was found to be excreted in the urine of man and rats and when the latter species was exposed to ionizing radiation, a procedure known to generate the ·OH in vivo (70), the quantity of cyclic 3-hydroxymelatonin increased incrementally in the urine. This provides direct evidence that when ·OH are generated in vivo, melatonin neutralizes them resulting in the formation of a metabolite that is excreted in the urine and is a footprint of ·OH generation (118). This will likely become a useful method for estimating in vivo ·OH production, something that has been difficult to access to date. The findings are also consistent with the repeated demonstration that melatonin reduces chromosomal damage induced by ionizing radiation (125, 126-128).
Besides scavenging the most toxic of the radicals produced, i.e., the ·OH, melatonin may also directly quench some other reactive oxygen metabolites. Singlet oxygen (1O2), which is formed by the addition of energy to ground state O2 (Fig. 1), although not a free radical, exhibits considerable toxicity and is capable of oxidizing polyunsaturated fatty acids and DNA (31). In vitro melatonin has been shown to reduce oxidative damage to neural lipids induced as a result of treatment of the animals with a photosensitizer, rose bengal, followed by the exposure of the neurons to bright light (20). Although this evidence is indirect, certainly the most likely explanation for melatonin's ability to reduce the products of lipid peroxidation and apoptosis in this model is its ability to quench 1O2 (86).
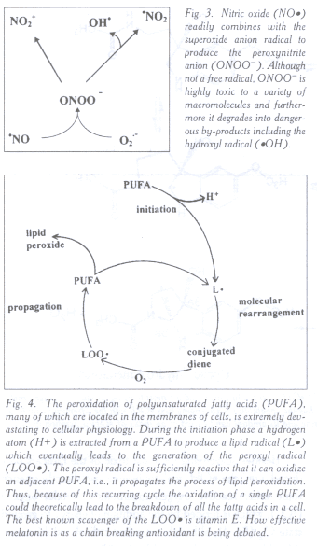
The peroxynitrite anion (ONOO-) is a non-radical but highly
toxic molecule that is formed from the combination of the superoxide
anion radical (O2•-) (Fig. 1) and nitric oxide (NO·)
(Fig. 3) (107). Besides its intrinsic toxicity, ONOO-
degrades into other agents that are equally or more highly toxic than
the parent molecule (91); one of these products may be the ·OH.
That melatonin effectively neutralizes the ONOO- was
recently shown by Gilad and co-workers (34). This group found that
the ability of ONOO- to oxidize dihydrorhodamine 123 was
reduced in the presence of melatonin in a dose-dependent manner.
They further found, using J774 macrophages, that melatonin reduced
DNA strand breaks and reversed the inhibition of mitochondrial
respiration resulting from ONOO- treatment. When compared
with other ONOO- scavengers, i.e., reduced glutathione and
cysteine, melatonin was found to be equally as effective. These
studies were further extended by Cuzzocrea et al. (26, 27) who
demonstrated that melatonin greatly reduced the inflammatory response
when animals are treated with carrageenan, where both ONOO-
and NO· are believed to be
the mediators of the inflammation (10). That melatonin scavenged the
ONOO- was demonstrated by a highly significant reduction
in the formation of nitrotyrosine, a footprint molecule in ONOO-
formation (26, 27).
NO· has both positive and negative actions in vivo. It is known to function as an important endothelium relaxing factor in small blood vessels and as a gaseous neurotransmitter in the brain (29). However, in high concentrations it may be responsible for the damaging effects of ischemia-reperfusion injury and, as noted above, it can combine with O2·- to produce the ONOO-, a molecule with well known toxicity. Besides scavenging ONOO-, as discussed above, Noda and colleagues (67) recently reported that melatonin directly scavenges NO· in vitro. While the significance of this finding has not yet been demonstrated in vivo, considering the widespread actions of NO·, especially in the central nervous system, the ability of melatonin to scavenge it may be significant particularly in terms of reducing its toxicity.
A major damaging agent of cellular physiology is the peroxyl radical (LOO·) which is generated during the peroxidation of lipids which are in high concentrations in cellular membranes (23). The LOO· has devastating consequences that are in part due to the fact that once formed during the process of lipid peroxidation it is sufficiently reactive so as to re-initiate (propagate) the processes; thus, theoretically at least once underway the process of lipid peroxidation would continue until all the lipid molecules in the membrane are oxidized (Fig. 4). This chain reaction can be interrupted by antioxidants such as vitamin E which readily scavenges the LOO· and protects against lipid peroxidation. Damaged lipids have marked consequences on membrane functions since they alter electrolyte transport, the activity of membrane-bound enzymes, channel functions, and membrane receptor processes. As the lipid moities of membranes become increasingly oxidized the membranes themselves become less fluid (32, 33). This rigidity of the membrane is a common feature of aging (29).
Melatonin's role in reducing lipid peroxidation is assured by the fact that it scavenges 1O2, ONOO- and ·OH, all of which are sufficiently reactive to initiate the peroxidative process. Evidence has also been presented, however, that melatonin functions as a chain breaking antioxidant by directly scavenging the LOO· (81, 82). Indeed, it was this group's conclusion that melatonin is twice as effective in scavenging the LOO· as is vitamin E, the primer chain-breaking antioxidant. This finding, if valid, would make melatonin the most effective chain breaking antioxidant discovered to date.
Confirmation of this finding, however, has not been forthcoming although several reports have shown that melatonin may have some LOO· scavenging activity. According to Scaiano (108), melatonin is probably equivalent to vitamin E in neutralizing the peroxyl radical. In studies by Marshall and colleagues (56) the high efficacy of melatonin in scavenging the LOO· as indicated by Pieri et al (81, 82) was not totally confirmed. The results of these reports are, however, difficult to directly compare since very different model systems were used to evaluate the ability of melatonin to scavenge the LOO·. A recent study also concluded that melatonin's ability to scavenge the LOO· is limited and probably not on a par with vitamin E (5).
While the efficacy of melatonin as a direct LOO· scavenge is debated, there is no doubt, especially in vivo, that it markedly inhibits lipid peroxidation due to a wide variety of toxins (21, 28, 63, 64, 89, 97). Whether this ability to limit the breakdown of lipids is solely the result of melatonin's ability to scavenge the initiating radicals or whether it also functions as a chain breaking antioxidant remains to be resolved. Besides reducing damaged lipid products in cellular membranes melatonin also prevents the changes in membrane fluidity which accompany the breakdown of polyunsaturated fatty acids (32, 33), thereby helping to preserve normal membrane and cellular physiology.
Besides the ability of melatonin to detoxify a variety of reactive, oxygen-based free radicals, it also indirectly reduces molecular damage by stimulating the metabolism of potentially toxic molecules to non-toxic products. At least two antioxidative enzymes metabolize H2O2, the precursor of the ·OH, to H2O. These enzymes are not equally distributed in cells but both catalytically remove H2O2 from the intracellular environment. One of these, glutathione peroxidase (GSH-Px) (Fig. 5) is selenium dependent and has been repeatedly shown to be stimulated by melatonin (8, 71, 72). When activated GSH-Px oxides reduced glutathione (GSH) to its disulfide form (GSSG) and in the process it uses H2O2 and other hydroperoxides as substrates, thereby reducing their concentrations. GSSG is converted back to GSH by the enzyme GSSG reductase (GSSG-Rd), a reaction that depends on the availability of NADPH. GSSG-Rd is also reportedly stimulated by melatonin ensuring that GSSG is recycled back to GSH (72). Finally, NADPH is replenished by the action of glucose-6-phosphate dehydrogenase (G-6PD), another antioxidative enzyme reported to increase its activity in response to melatonin (84). Besides removing H2O2 from cells, GSH-Px also functions as a peroxynitrite reductase thereby metabolizing ONOO- to a non-toxic product.
A major antioxidative family of enzymes which metabolizes O2·- are the superoxide dismutases (SOD). These rapidly convert O2·- to H2O2 thereby restricting the interaction of O2·- with NO· and limiting the inherent toxicity of O2·-. Melatonin has now been shown to stimulate both mRNA for SOD as well as increasing the activity of the enzyme (4, 48). In so doing, melatonin would be expected to increase the intracellular concentrations of H2O2 which would normally enhance the formation of the highly toxic ·OH [this is what happens normally in individuals with trisomy 21 (Down syndrome) where SOD activity is increased 1.5-fold]. However, besides stimulating SOD activity, as already mentioned, melatonin also augments GSH-Px activity and reduces H2O2 thereby decreasing the likelihood of ·OH formation by concurrently increasing the formation of H2O2 from O2·- and augmenting its metabolism to non-toxic products. In this way melatonin can avert the macromolecular damage which results when only SOD activity is increased. There is also one potentially pro-oxidative enzyme, nitric oxide synthase (NOS), which is inhibited by melatonin. As noted above NO, which is synthesized from arginine under the influence of NOS, has some intrinsic toxicity and furthermore it degrades into the ONOO- (when it combines with O2·-). Thus, the formation of NO is sometimes a pro-oxidative process. At both physiological and pharmacological concentrations in vivo melatonin is known to inhibit neuronal NOS activity and consequentially the accumulation of excessive NO (13, 87, 88). This action of melatonin contributes to the indirect means by which the indole is capable of reducing oxidative stress, at least in the central nervous system.
Age-related Changes in Melatonin
While there may be a variety of organs that synthesize melatonin, the concentrations of this indole in the blood are usually considered to be primarily or wholly derived from the pineal gland under usual conditions (92). This is quite remarkable since the retinas also produce melatonin in a rhythm reminiscent of that seen in the pineal gland, but unlike the pineal, they are incapable of releasing the synthetic product or it is locally metabolized before it is released.
In the pineal gland, melatonin is produced almost exclusively during the night and it seems to be immediately released after its synthesis resulting in nocturnal blood levels that are substantially higher at night than during the day (92). This rhythmic pattern of melatonin production and release is common to all vertebrates. While the rhythm is typically not present in newborns, it appears soon thereafter. From infancy to middle-aged the melatonin rhythm is detectable in the blood of mammals although in humans their may be large variations in the nocturnal melatonin increase even among individuals of the same age (6).
Absolute levels of melatonin in the blood are usually measured in pg•ml-l with these values being below 20 pg•ml-I during the day up to 150-200 pg•ml-l at night. Relatively little is known about intracellular concentrations of melatonin although under some circumstances these values can exceed those measured in the blood by a wide margin (62). The low values of melatonin in the blood would seem to detract from the possibility that physiological concentrations of the indole are relevant in terms of the total antioxidative capacity of the organism. When the total antioxidant status of the serum of rats and humans are compared with the concentration of melatonin in the same samples, however, there is a very high correlation (11, 12). This is consistent with the observations that endogenously produced melatonin is sufficient to provide partial protection against agents or processes which greatly increase oxidative damage (55, 120). Thus, the antioxidative capacity of melatonin seems not to be a phenomenon exclusively related to the administration of pharmacological concentrations of the indole, but rather the amount of melatonin produced endogenously is important in providing significant protection against the devastating actions of free radicals.
In light of the seemingly relative importance of endogenously produced melatonin
in terms of antioxidative defense, depression in melatonin production
due to any cause could be significant relative to protection from
free radicals. One factor that leads to a reduction in melatonin is
aging (93). In every mammalian species where the melatonin synthetic
and secretory capability of the pineal gland have been measured they
diminish in advanced age and this is
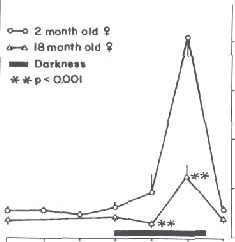
Time associated with low levels of melatonin in the blood even at night. Of the mammals studied to date the rat (75, 98), Syrian hamster (74, 101) (Fig. 6) and Mongolian gerbil (99) have all exhibited a reduction in the pineal synthesis of melatonin after middle age. Similar studies in humans using either blood levels of melatonin or the excretion of the chief enzymatic metabolite of melatonin, 6-hydroxymelatonin sulfate, as endpoints have yielded similar findings (41, 66, 106, 123). The conclusion from these studies is that elderly individuals produce less pineal melatonin and, due to the loss of this antioxidant, they are increasingly vulnerable to free radical damage. The drop in melatonin in advanced age may come at a highly non-propitious time since it has been shown that free radical generation increases in aged individuals (129). Thus in a since melatonin may be lost when it is needed most. Whether in fact the reduced melatonin production is consequential in terms of the obvious age-associated, free radical-based diseases that commonly occur simultaneously is unknown but is being actively investigated. Furthermore, although the suggestion has been made that secretory products of the pineal gland may be able to defer aging per se (69, 83) the experimental data are in complete and, at this point, not compelling (100, 102).
Melatonin and Age-related Diseases
There are a host of age-related diseases that are in part related to free radical damage and, therefore, could possibly be deferred by the use of a proper antioxidant regimen. Only a small number of these diseases will be considered here. One of the most common surgical procedures performed in the elderly is the replacement of the opaque lenses of the eyes. The opaqueness, i.e., cataracts, are classical free radical damage to macromolecules in the lens which leads to molecular deterioration and cloudiness causing these individuals to loose their sight (43). Since cataracts are free radical-based, one may anticipate that their development would be deferred by antioxidants. We have in fact tested an animal model of cataracts and found that indeed melatonin can prevent experimental cataract induction (1, 51). In this model newborn rats are treated with the drug buthionine sulfoximine (BSO) which inhibits gamma-glutamyl cysteine synthase, the rate limiting enzyme in the production of an important antioxidant, reduced glutathione (GSH). As a result of this treatment the animals develop grossly observable cataracts by the time they are 2 weeks of age, i.e., several days after their eye lids open. Newborn rats, however, produce very little melatonin (at least in their pineal gland) during the first 2 weeks of life. Thus, in the experiment described, the newborn rats were actually deficient in two important antioxidants, GSH and melatonin. This being the case, we surmised that if newborn rats who developed cataracts because they were GSH deficient (and melatonin deficient) were given melatonin daily during the experimental period, the cataracts would not form. This predicted outcome was in fact realized. Melatonin given as a single daily injection prevented the development of cataracts in newborn rats that had been treated with BSO to deplete their GSH. Thus, the antioxidant melatonin had adequately substituted for the antioxidant, GSH thereby preventing free radical damage to macromolecules in the lens and inhibiting the development of cataracts (1, 51).
On the basis of these studies, it may be theorized that cataractogenesis in humans, which typically occurs in advanced age when endogenous melatonin levels are depleted, may be related to the loss of this important antioxidant. Since no tests have yet been performed to examine the effects on melatonin on human cataractogenesis, however, the assumption that opaquification of the human lens is related to the reduction in melatonin remains precisely that, an assumption, and would seem to be worthy of examination.
Many types of cancer increase in frequency as individuals age. In many cases, the initiating events, i.e., the damage to nuclear DNA, is mediated by free radicals (3, 52). If the damaged DNA undergoes mutation, cancer can be a consequence. Not only are free radicals often involved in the initiation of cancer but their promotion may also involve free radicals (22). Finally, recent evidence suggests that the transformation of benign cancers to their more malignant forms may relate to the continuing oxidative damage sustained by nuclear DNA (54).
Since melatonin has been found in the nucleus (30, 62), it is in a position to scavenge free radicals that could potentially damage DNA. The initial studies which examined melatonin's ability to reduce oxidative damage to DNA were published roughly 5 years ago (119, 120). In this case, rats were treated with the chemical carcinogen safrole with and without concurrent melatonin administration. When damaged DNA adducts were measured in the liver of these rats, melatonin had proven highly effective in reducing the quantity of DNA adducts that had formed. The presumed mechanism of the reduced DNA damage related to the ability of melatonin to detoxify the free radicals generated during the metabolism of the carcinogen before they mutilated the nearby DNA.
If in fact melatonin is an effective ·OH scavenger as has been claimed, one would expect it to protect against genomic damage that accompanies the exposure of animals to ionizing radiation. Ionizing radiation is known to cause the homolytic scission of H2O molecules resulting in the formation of ·OH which then go on to damage DNA as well as other cellular macromolecules (70). In all experiments performed to date, melatonin has proven highly effective in reducing cytogenetic damage in cells exposed to either ionizing radiation (125-128) or radiomimetics (7, 65). Furthermore, the death rate in mice given a lethal dose of radiation has been shown to be reduced when animals are pretreated with melatonin (124). These findings are consistent with melatonin being an effective radioprotective agent in part related to its ability to curtail oxidative damage to nuclear DNA. In so doing, melatonin would also be expected to suppress cancer initiation. Using the same endpoints to assess DNA damage, Sewerynek and colleagues (110) and Melchiorri et al. (60) have shown that melatonin also protects against the genotoxic actions of bacterial lipopolysaccharide and paraquat, respectively.
Still other scientists have used other techniques to estimate the efficacy of melatonin in protecting against oxidative damage to DNA. According to Susa and colleagues (116), DNA strand breaks normally associated with the exposure of hepatocytes to chromium (VI) were prevented when melatonin was also present in the incubation medium. In this case the authors concluded that melatonin was a potent protector of DNA against such damage. Finally, using measures of 8-hydroxydeoxyguanosine (8OHdG), a documented damaged DNA product, Tang and co-workers (121) found melatonin to reduce in vivo 8OHdG levels in rat liver and brain after their treatment with the free radical generating agent kainic acid.
Again, these studies like those summarized in the preceding paragraphs, suggest that melatonin would decrease the incidence of cancer which is initiated as a result of free radical damage. Heretofore it has been assumed that melatonin's protective effect against cancer related to its ability to reduce the growth of growing cancers, a phenomenon that has been thoroughly investigated (14, 76). In light of the data summarized above, however, it seems that reducing both the initiation and promotion of tumors may be within the capability of melatonin. These findings should then be considered in the context of the aged individual where endogenous melatonin production may be minimal.
Neurodegenerative diseases occur more frequently now than earlier in the century. The major reason for this presumably is that humans are living progressively longer allowing these diseases of the aged to become manifested. This being the case, it is anticipated that the prevalence of these conditions will continue to rise inasmuch as people are surviving longer and longer. While neurodegenerative conditions often have complex etiologies there is widespread agreement that many of the disease processes involve free radicals (Table 2). In the current review only the association of melatonin with models of Alzheimer's disease, Huntington's disease and Parkinsonism will be considered.
Alzheimer's disease, formally referred to as presenile dementia, afflicts
approximately one of 20 people over 65 years of age while by 85 this
increases to one in three individuals. This disorder is extremely
debilitating and is characterized by a gradual decline in virtually
all brain functions including memory, judgement, behavior,
personality, abstract thinking, language and motor skills. The
deterioration can be slow (over a 7-10 year period) and less
frequently rapid. In the latter stages of the disease the subjects
often have a feeling of anxiety, restlessness and agitation in the
later afternoon, a collection of signs referred to as sundowning. A
large amount of research suggests that the deposition of a product
called
In a series of two reports we (79, 80) we found that melatonin readily
inhibited apoptosis of neurons incubated in the presence of either
the 25-35 amino acid residue of
Besides reducing the toxicity of
A couple of reports have appeared where melatonin has actually been given as a supplement to individuals suffering with Alzheimer's disease. The studies included a rather small number of subjects and they were not conducted in a double-blind manner. Significantly, however, both of these studies indicated that giving as little as 6 mg melatonin daily over a 36 month period delayed the progression of the disease, based on their behavioral assessment on standardized tests, and improved the general well being of the Alzheimer's patients (16, 17).
Huntington's disease (or Huntington's chorea) is an autosomal dominant condition in which neurodegeneration is a major feature (122). This disease usually first manifests itself in middle-aged individuals and is characterized by motor disturbances, behavioral changes and dementia. Commonly, brain weight may be reduced by 20% when these individuals die (122).
A causative factor for this debilitating disease is believed to be the excitotoxic tryptophan metabolite, quinolinic acid (9). In the study in question, quinolinic acid was injected directly into the striatum of the rat, a treatment which reproduced the neuropathyology observed in the brains of individuals' who died with Huntington's disease. These observations have led to the wide spread use of quinolinic acid administration as an experimental model for this condition. As an excitatory neurotoxin, quinolinic acid is believed to damage and kill neurons via free radical mechanisms after it interacts with the N-methyl-D-aspartate (NMDA) receptor. It is especially damaging since it is not metabolized and therefore it acts on the receptors for a prolonged period (115).
Given that free radicals are the presumed destructive neuronal agents in the Huntington's disease model, Southgate and colleagues (113) assumed that melatonin may provide protection against the toxicity of quinolinic acid much like it did in the models of Alzheimer's disease. After the injection of quinolinic acid directly into the hippocampus of adult rats, the excitotoxin caused distinctive morphological changes in the pyramidal neurons with 5 days. Additionally, they measured a reduction in glutamate receptor number in the hippocampus following quinolinic acid administration. When melatonin was given just prior to excitotoxin administration, neither the morphological changes nor the alterations in glutamate receptor numbers were apparent; the authors theorized that melatonin's antioxidant activity accounted for its ability to protect against quinolinic acid.
Although the cause of Parkinson's disease remains to be clarified, it is clearly age-related with an age of onset being between 40-65 years. This disorder is characterized by tremor, rigidity, slowness of movement and difficulty in walking. That oxygen free radicals are involved in the destruction of neurons in individuals with Parkinsonism is now widely accepted. The neurons that exhibit the most marked reduction are the dopaminergic neurons of the pars compacta of the substantial nigra.
For the experimental induction of Parkinson's disease, 1-methyl-4-phenyl-1,2,3,4-tetrahydropyridine (MPTP) is the agent most frequently used. After its uptake by glial cells, MPTP is metabolized to the methyl-4-phenyl pyridinium cation (MPP+) which is then released and taken up by dopaminergic neurons where it generates destructive free radicals. This model has been used by Acuna-Castroviejo et al (2) to test the efficacy of melatonin in curtailing the toxicity of MPTP. Melatonin was found to limit the loss of dopamine and the reduction of striatal tyrosine hydroxylase activity observed in the MPTP-treated mice not given melatonin. Likewise, the neural lipid peroxidation induced by MPTP was prevented by melatonin. These findings are consistent with the antioxidative actions of the indole. When the same biochemical endpoints were measured, Kim et al. (45) also showed that melatonin reduced the damage as well as the biobehavioral changes in rats treated with 6-hydroxydopamine (6OHDA); this agent also destroys dopaminergic neurons and induces Parkinson-like signs in experimental animals.
Besides the in vivo studies described above, two reports that used in vitro methods have confirmed the neuroprotective effects of melatonin against MPP+ and 6OHDA toxicity (40, 59). Both these studies monitored similar endpoints and concluded that melatonin's protective actions most likely related to its ability to detoxify free radicals induced by the respective toxins.
Concluding Remarks
This review is by no means exhaustive in terms of agents and processes, all of which are believed to involve free radicals, against which melatonin has been found to be protective. Rather, the examples summarized herein were chosen to illustrate how melatonin may relate to the progression of age-related diseases and to aging itself. It is obvious from the reports discussed herein that the bulk of the studies have involved experimental animals and that the number of clinical studies is small. Considering the overwhelmingly positive nature of melatonin's protective actions in these experimental studies, it would seem clinical trials are now warranted. This would seem justified considering the virtual absence of toxicity of melatonin and its apparent numerous beneficial effects. Furthermore, several of the diseases against which it could be tested have no known effective treatments. Finally, the importance of these studies is further emphasized by the fact that humans are surviving progressively longer and, as a result, some of the most devastating age-related diseases will continue to increase in frequency. Unless successful treatments can be found for these conditions the burden to society, both from the standpoint of effective care as well as financially, may become insurmountable. Studies to date indicate melatonin may be able to relieve part of this burden.
References
Fig. 2. Melatonin is a highly efficient scavenger of the hydroxyl radical (·OH), generally considered the most toxic by-product of oxygen metabolism. We have recently shown, in fact, that each melatonin molecule has the capability of scavenging two ·OH with the "footprint" molecule being cyclic 3-hydroxymelatonin. This product is then excreted in the urine and its amount can be used as an index of ·OH generation in vivo.
Fig. 3. Nitric oxide (NO·) readily combines with the superoxide anion radical to produce the peroxynitrite anion (ONOO-). Although not a free radical, ONOO- is highly toxic to a variety of macromolecules and furthermore it degrades into dangerous by-products including the hydroxyl radical (·OH).
Fig. 4. The peroxidation of polyunsaturated fatty acids (PUFA), many of which are located in the membranes of cells, is extremely devastating to cellular physiology. During the initiation phase a hydrogen atom (H+) is extracted from a PUFA to produce a lipid radical (L·) which eventually leads to the generation of the peroxyl radical (LOO·). The peroxyl radical is sufficiently reactive that it can oxidize an adjacent PUFA, i.e., it propagates the process of lipid peroxidation. Thus, because of this recurring cycle the oxidation of a single PUFA could theoretically lead to the breakdown of all the fatty acids in a cell. The best known scavenger of the LOO· is vitamin E. How effective melatonin is as a chain breaking antioxidant is being debated.
Fig. 5. Hydrogen peroxide (H2O2), while not being a free radical, is highly dangerous because it degrades into the hydroxyl radical (·OH) (see Fig. 1). Fortunately, much of the H2O2 generated is metabolized to H2O by the actions of two detoxifying enzymes, catalase and glutathione peroxidase. Melatonin has been shown to stimulate the activity of glutathione peroxidase which converts reduced glutathione (GSH) to its oxidized form (GSSG). GSSG is catalytically metabolized back to GSH by the enzyme glutathione reductase, which is also reportedly stimulated by melatonin. A major source of H2O2 in cells is produced by the dismutation of the superoxide anion radical (O2-·), a conversion that requires one of a family of superoxide dismutases (SOD).
Fig. 6. In those mammals where it has been studied, pineal melatonin production diminishes with increased age. Shown here are the pineal melatonin rhythms in 2 month old and 18 month old female Syrian hamsters. Clearly, in the old animals the nocturnal peak of melatonin is attenuated. A similar reduction in pineal melatonin production has been observed in the human. Table 1. Calculated rate constants (Kr) for the scavenging of the ·OH by melatonin and related methoxylated and hydroxylated indoles. Data from Tan et al. (1993), Poeggeler and colleagues (1996) and Matuszek et al. (1997).
Table 2. Neurodegenerative conditions, most of which are associated with the aging population, are believed in many cases to involve free radicals as part of the disease process. It should be noted that whereas the following list of conditions can be at least in part ameliorated by appropriate free radical scavengers and antioxidants, it is not likely that a single antioxidant will totally prevent any of these conditions.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Размещение рекламы | |
|
| |

 Articles
Articles